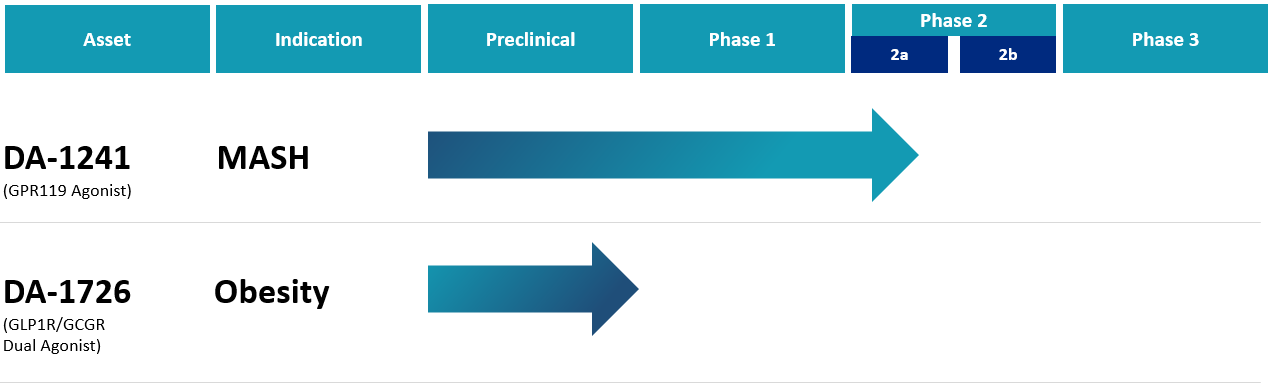
MASH is a silently progressing metabolic liver disease in which the liver has more than 5% fat and has varying degrees of inflammation and fibrosis. Cardiovascular disease (CVD) is a leading cause of morbidity and mortality in MASH patients. As metabolic diseases characterized by calorie excess, particularly diabetes and obesity, become more common worldwide, the incidence of MASH is increasing. Long-term persistence of hyperglycemia and/or hyperlipidemia increase the incidence of cardiovascular disease. Therefore, improving liver lesions and efficiently controlling comorbidities in MASH patients are important for effectively reducing the risk of cardiovascular events. To date, there are no anti-MASH drugs approved by the FDA.
DA-1241 is a new chemical agent activating G protein-coupled receptor 119 (GPR119) mainly in the pancreas, intestine, and liver. In non-clinical studies, GPR119 activation by DA-1241 in hepatocytes, macrophages, and hepatic stellate cells inhibits lipid accumulation, immune cell infiltration, and the production of collagen fibers in the liver, directly ameliorating MASH pathophysiology such as steatosis, inflammation, and fibrosis. Moreover, GPR119 has a distinctive role in glucose and lipid metabolism via stimulating secretion of insulin and glucagon-like peptide-1 (GLP-1) in pancreatic beta cells and intestinal L-cells, respectively. Therefore, DA-1241 can also provide additional metabolic benefits in MASH patients with common comorbid metabolic diseases such as type 2 diabetes and dyslipidemia.
In Phase 1b clinical trials, the safety and efficacy of DA-1241 monotherapy were successfully confirmed. DA-1241 was well-tolerated in type 2 diabetic patients. GPR119 activation by DA-1241 treatment increased the plasma levels of gut peptide hormones (total GLP-1, PYY, GIP) as plasma concentrations of DA-1241 went up and showed a persistent and similar postprandial glucose-lowering activity to sitagliptin (JANUVIA®) (an oral anti-diabetic agent as a dipeptidyl peptidase inhibitor) in a head-to-head trial after 8 weeks of once daily oral treatment.
